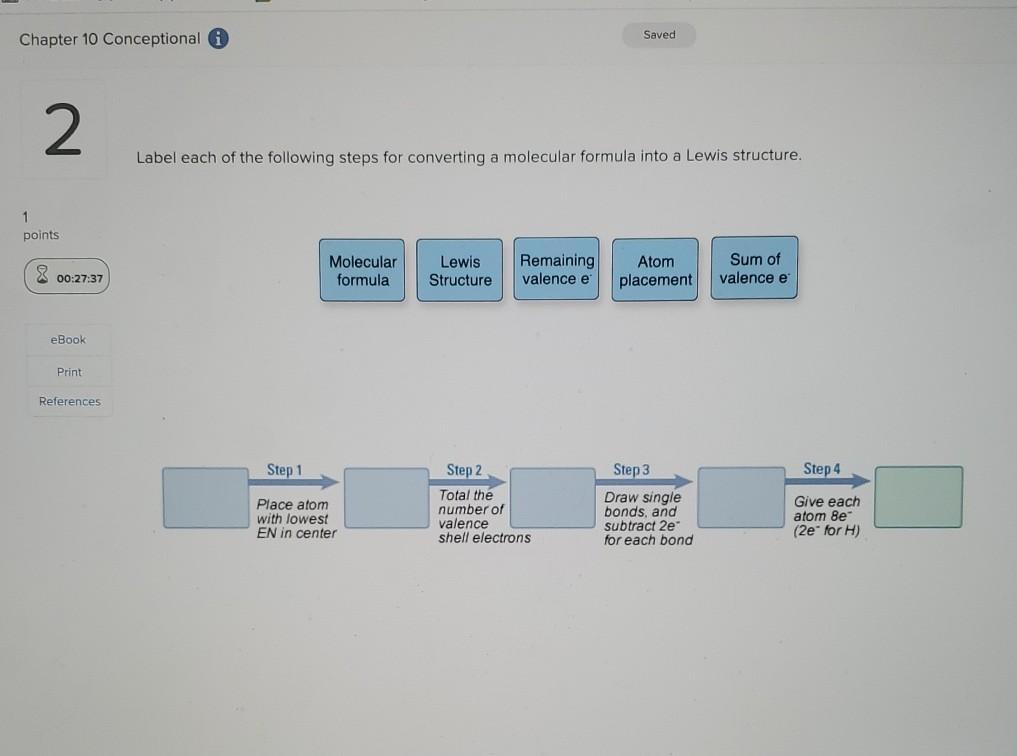
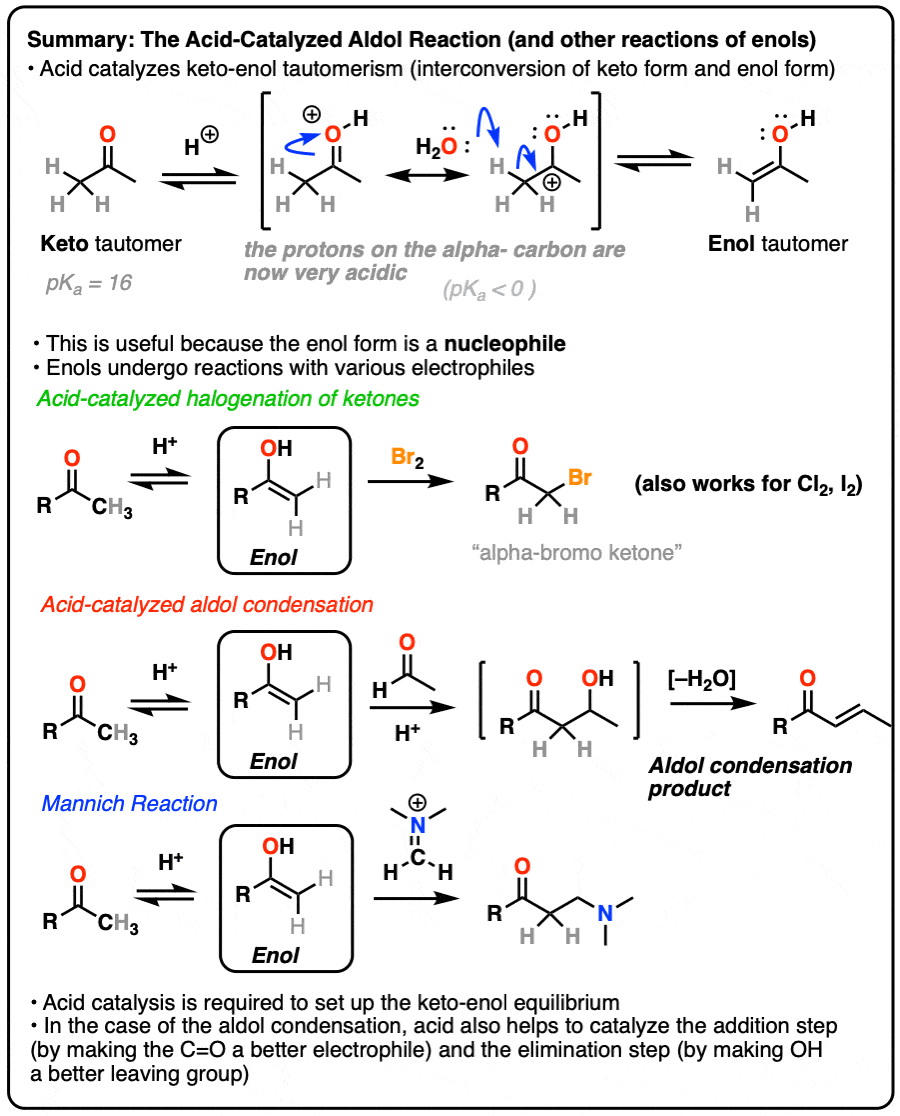
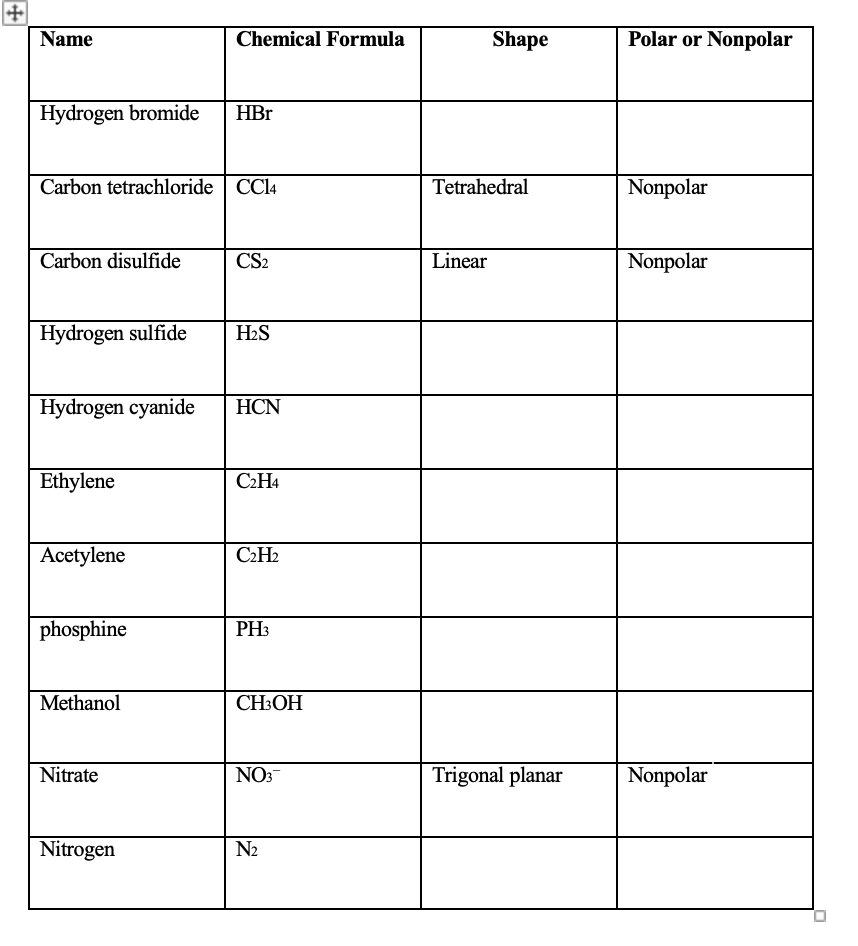
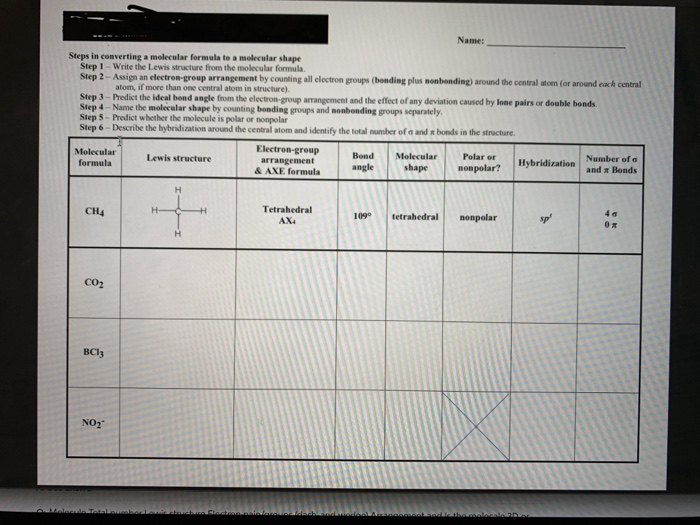
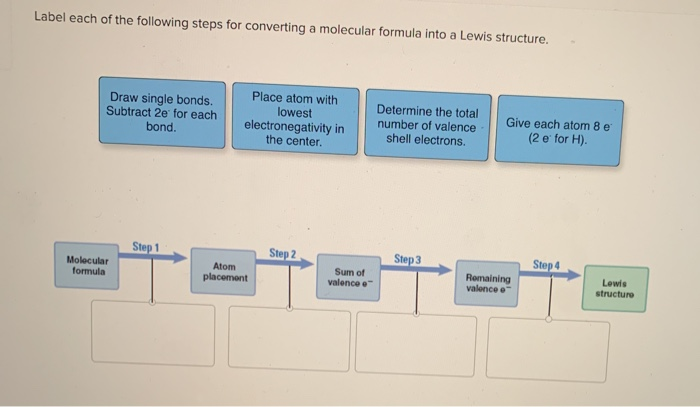
39 label each of the following steps for converting a molecular formula into a lewis structure.
Lewis Symbols and Structures | Chemistry for Majors - Lumen Learning To draw the Lewis structure for an odd-electron molecule like NO, we follow the same six steps we would for other molecules, but with a few minor changes: Determine the total number of valence (outer shell) electrons. The sum of the valence electrons is 5 (from N) + 6 (from O) = 11. How to Find Molecular Formula: 13 Steps (with Pictures) - WikiHow Multiply the subscripts of the empirical formula by the ratio. This will yield the molecular formula. Note that for any compound with a ratio of "1," the empirical formula and molecular formula will be the same. Example: C12OH30 * 2 = C24O2H60 Part 2 Finding the Empirical Formula Download Article 1 Find the mass of each atom present.
Steps to convert molecular formula into a Lewis Structure. - Quizlet Study with Quizlet and memorize flashcards containing terms like Step 1, Step 2, Step 3 and more. ... Subjects. Expert solutions. Log in. Sign up. Chapter 10 - Steps to convert molecular formula into a Lewis Structure. Flashcards. Learn. Test. Match. Step 1. ... Give each atom 8e- (2e- for H). Other sets by this creator. Ch6 Vocab Quiz. 46 ...
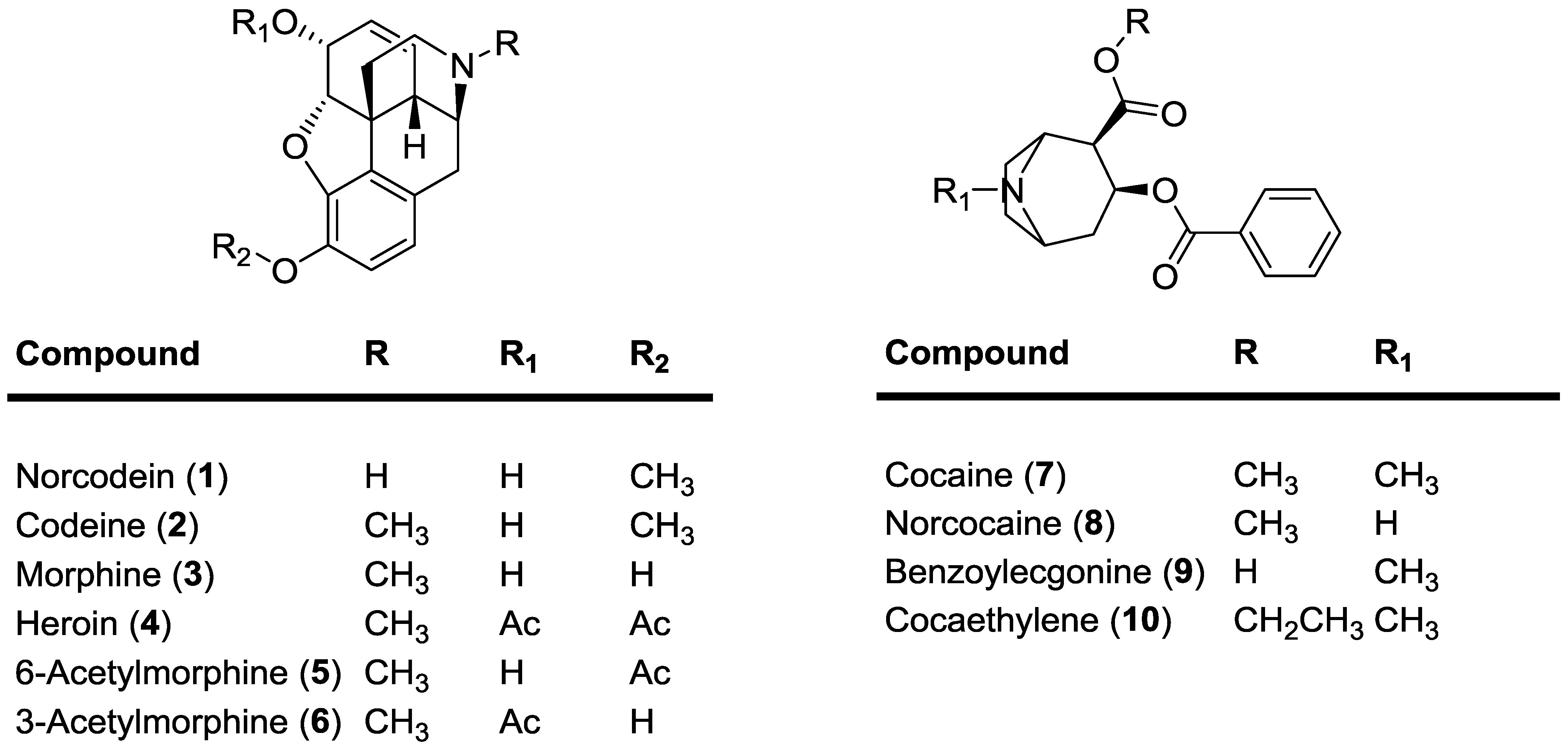
Label each of the following steps for converting a molecular formula into a lewis structure.
Drawing Lewis diagrams (video) | Khan Academy A Lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. 4.4: Drawing Lewis Structures - Chemistry LibreTexts For a molecule, we add the number of valence electrons (use the main group number) on each atom in the molecule. This is the total number of electrons that must be used in the Lewis structure. O + 2 (F) = OF 2 6e - + (2 x 7e -) = 20e - H + C + N = HCN 1e - + 4e - + 5e - = 10e - 2. 9.3: Drawing Lewis Structures - Chemistry LibreTexts Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure."
Label each of the following steps for converting a molecular formula into a lewis structure.. Lewis Electron Dot Structures - Detailed Explanation with Examples & Videos First, the total number of valence electrons present in the molecule is calculated by adding the individual valencies of each atom. If the molecule is an anion, extra electrons (number of electrons added = the magnitude of negative charge) are added to the Lewis dot structure. How to Draw a Lewis Structure - ThoughtCo Step 1: Find the Total Number of Valence Electrons In this step, add up the total number of valence electrons from all the atoms in the molecule. Step 2: Find the Number of Electrons Needed to Make the Atoms "Happy" An atom is considered "happy" when its outer electron shell is filled. Label each of the following steps for converting a molecular formula ... The following procedure can be used to construct Lewis electron structures for more complex molecules and ions: How-to: Constructing Lewis electron structures. 1. Determine the total number of valence electrons in the molecule or ion. Add together the valence electrons from each atom. How to Write a Chemical Formula Given a Chemical Structure Writing a Chemical Formula Given a Chemical Structure Example 1. Write the chemical formula for the molecule depicted by the following chemical structure. Step 1: Identify the elements in the ...
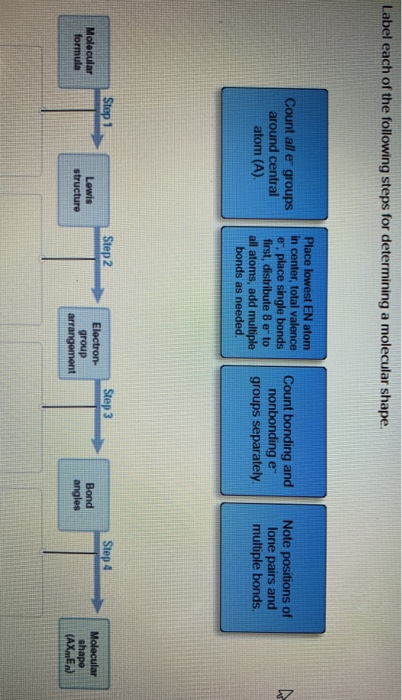
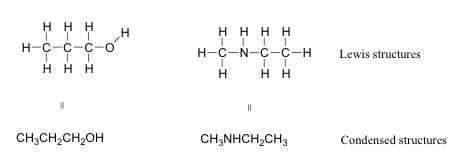
Bond-line, Lewis and Condensed Structures with ... - Chemistry Steps Follow these steps: 1) Number all the atoms (except for H's). Note: Numbering atoms will help you immensely in doing any transformation in Organic Chemistry. It does not have to be in a particular order or follow the IUPAC rules. 2) Simply connect all the atoms from left to right. Note that some connections may be wrong at this point. Lewis structure calculator | Lewis structure generator To use the Lewis Structure Calculator follow these steps: Enter the formula of the molecule in the field provided for it. For example, if we want to obtain the Lewis structure of the Sulfate ion, SO 4 - 2, we must first enter the charge by typing (-2) or by entering -2 in the charge field and pressing the «Add» button. Then we write the rest of the formula being as follows: (-2)SO4. chem PROCTOR Flashcards | Quizlet label each of the following steps for determining a molecular shape 1.) molecular formula 2.) lewis structure 3.) electron group arrangement 4.) bund angles label each of the following steps for determining a molecular shape pt. 2 1.) (Ions Only): ionic bonding 2.) ( Ions + Polar Molecules): ion-dipole forces 3.) How To: Drawing Lewis Structures From Condensed Molecular Formulas There are three steps you should follow to draw a correct structure. 1. From a condensed molecular formula, you obtain information about which atoms are connected to each other in a molecule. Connect all of the appropriate atoms with single bonds first (lines). Example: CH3CH2CH2CO2CH3
Lewis Structures: Dot Symbols, How to Draw, Significance - Embibe The Lewis structure was named after Gilbert N. Lewis, who introduced it in his \ (1916\) article "The Atom and the Molecule .". In a Lewis Structure, electrons are represented as "dots" surrounding the central metal atom. The central metal is denoted by using its chemical symbol from the Periodic Table. Solved Label each of the following steps for converting a - Chegg Molecular Remainin Structureformula g valence Lewis Sum of Atom valence eplacement Step 1 Step 2 Step 3 Step 4 Place atom with lowest EN in center Total the number of valence shell electrons Draw single bonds, and subtract 2e for each bond Give each atom 8e (2e for Show transcribed image text Expert Answer 95% (73 ratings) Lewis Structures: Learn How to Draw Lewis Structures | Albert.io Examples for Drawing Lewis Structures for Covalent Bonds . Here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. Reference the "How to Draw a Lewis Dot Structure" for a Step by Step guide. See the following Lewis dot structure diagrams for a few covalent compounds. Example 1. Solved Label each of the following steps for converting a - Chegg Question: Label each of the following steps for converting a molecular formula into a Lewis structure. Draw single bonds. Subtract 2e for each bond. Place atom with lowest electronegativity in the center. Determine the total number of valence shell electrons. Give each atom 8 e (2 e for H).
label each of the following steps for converting a molecular formula ... Label steps converting following lewis structure each molecular formula solved into step transcribed problem text been. 34 label each of the following steps for converting a molecular formula. ← emo anime guy Stylish zone: sad emo boys coolest anime backgrounds Best minecraft backgrounds ...
2 Label each of the following steps for ... - Inorganic Chemistry 2 Label each of the following steps for converting a molecular formula into a Lewis structure. 3.03 points Place atom with lowest electronegativity in the center. Determine the total number of valence shell electrons. Draw single bonds. Subtract 2e for each Give each atom 8 e (2 e for H). eBook bond.
6.1 Lewis Electron Dot Diagrams | Introductory Chemistry - Lumen Learning A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the ...
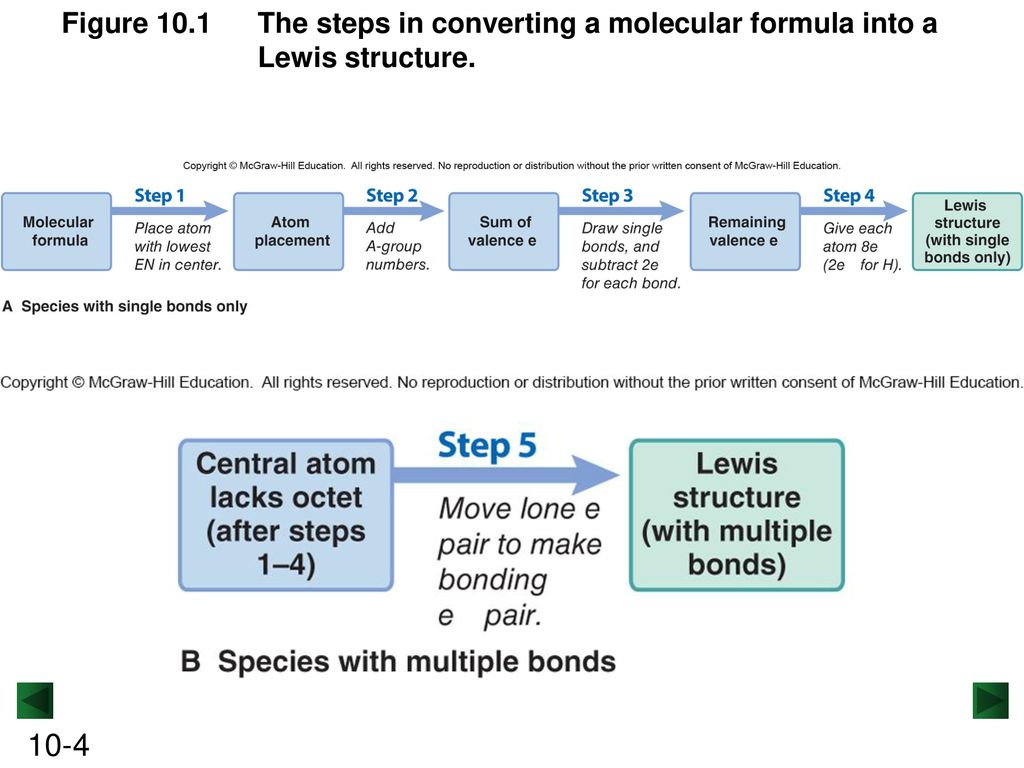
PDF Chapter 10 The steps in converting a molecular formula into a Lewis structure. Molecular Formula Atom placement Place atom with lowest EN in center. Step 1 Step 2 Add A -group numbers. Sum of valence e Draw single bonds, and subtract 2e for each bond. Step 3 Remaining valence e Lewis structure Step 4 Give each atom 8e (2e for H). 10-4 Example: NF 3
Molecular Formula: Definition & Calculate Empirical Formula - Embibe Exams The following steps can determine the molecule formula of a compound- 1st Step: Calculate the empirical formula from percentage composition. 2nd Step: Calculate the Empirical Formula mass (EFM) by adding up the molar atomic masses of all atoms constituting the formula. 3rd Step: Determination of the Molar Mass of the given compound.
label each of the following steps for converting a molecular formula ... Label steps converting following lewis structure each molecular formula solved into step transcribed problem text been. 34 label each of the following steps for converting a molecular formula. ← transparent natural light logo Free colorful line cliparts, ...
label each of the following steps for converting a molecular formula ... Get the detailed answer: label each of the following steps for converting a molecular formula into a lewis structure.
9.3: Drawing Lewis Structures - Chemistry LibreTexts Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure."
4.4: Drawing Lewis Structures - Chemistry LibreTexts For a molecule, we add the number of valence electrons (use the main group number) on each atom in the molecule. This is the total number of electrons that must be used in the Lewis structure. O + 2 (F) = OF 2 6e - + (2 x 7e -) = 20e - H + C + N = HCN 1e - + 4e - + 5e - = 10e - 2.
Drawing Lewis diagrams (video) | Khan Academy A Lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms.


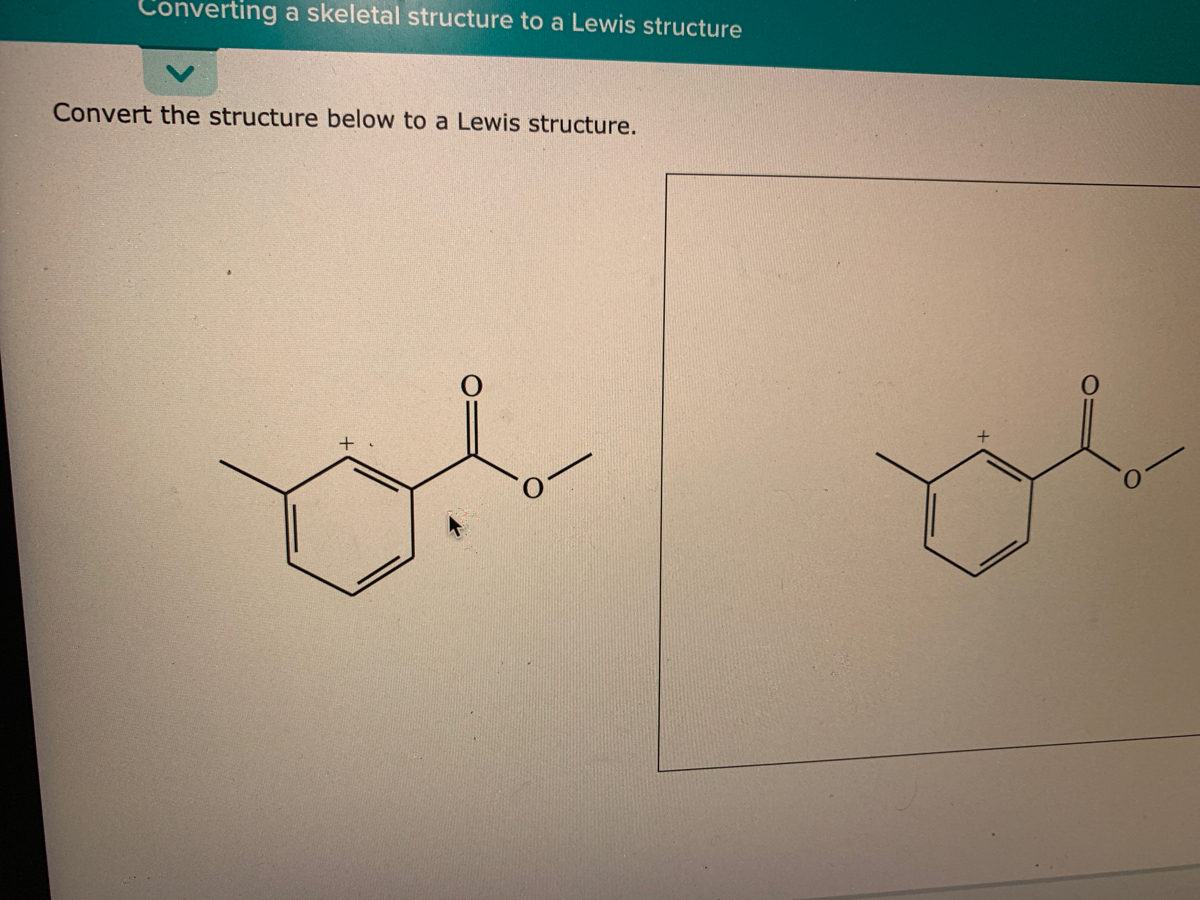









:max_bytes(150000):strip_icc()/lewisnitrite-56a128825f9b58b7d0bc90cf.jpg)



Post a Comment for "39 label each of the following steps for converting a molecular formula into a lewis structure."